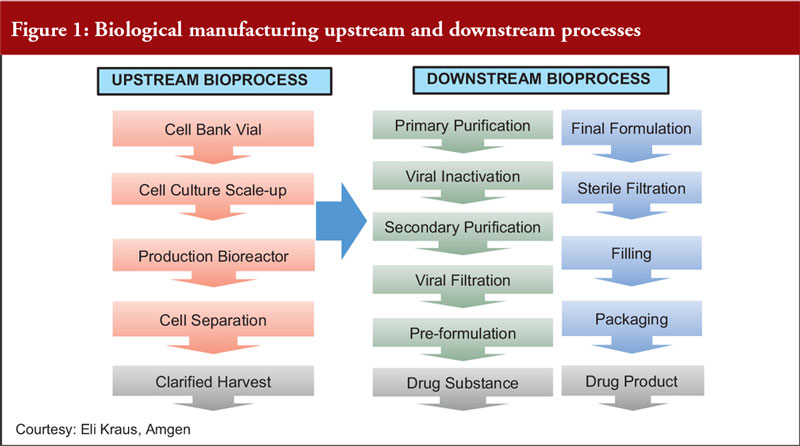
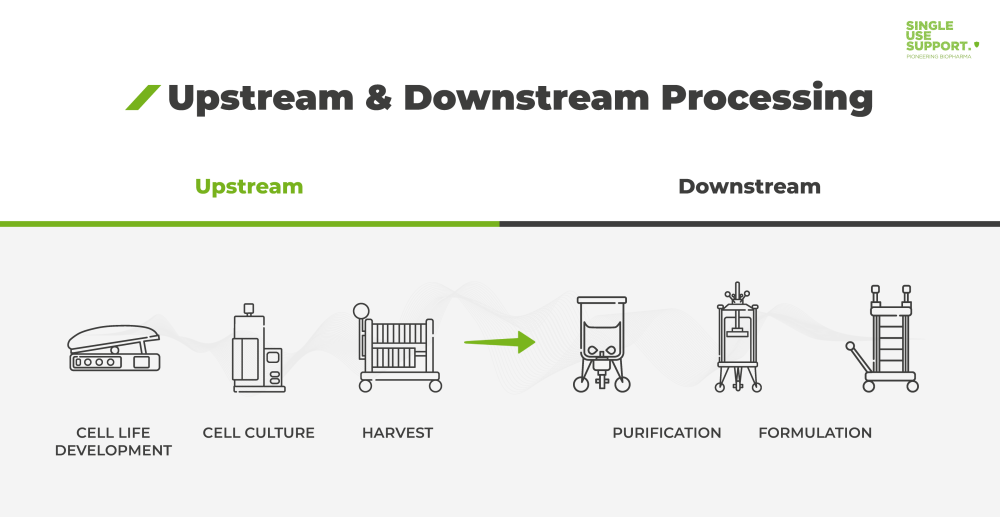
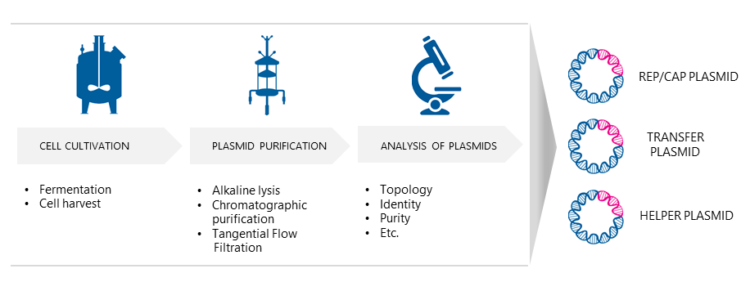
Phase-Dependent Approaches to Plasmid DNA Manufacture: Cobra Biologics describes how new guidelines for starting materials benefit developers of vectors for gene-based advanced therapy medicinal products: Genetic Engineering & Biotechnology News: Vol 41,

Figure 16 from Mise en place et développement d'une plateforme de culture pour la levure pichia pastoris | Semantic Scholar
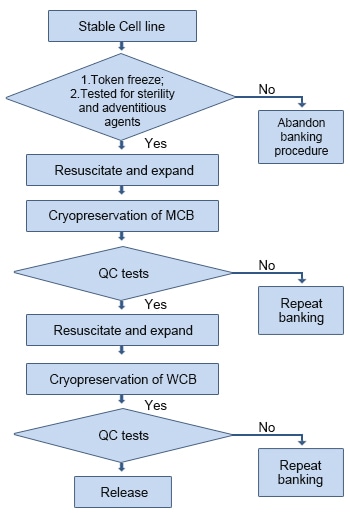
CELL BANKS PREPARATION IN BIOPHARMACEUTICALS PRODUCTION/ TWORZENIE BANKOW KOMOREK W PRODUKCJI BIOFARMACEUTYKOW. - Document - Gale Academic OneFile
CELL BANKS PREPARATION IN BIOPHARMACEUTICALS PRODUCTION/ TWORZENIE BANKOW KOMOREK W PRODUKCJI BIOFARMACEUTYKOW. - Document - Gale Academic OneFile

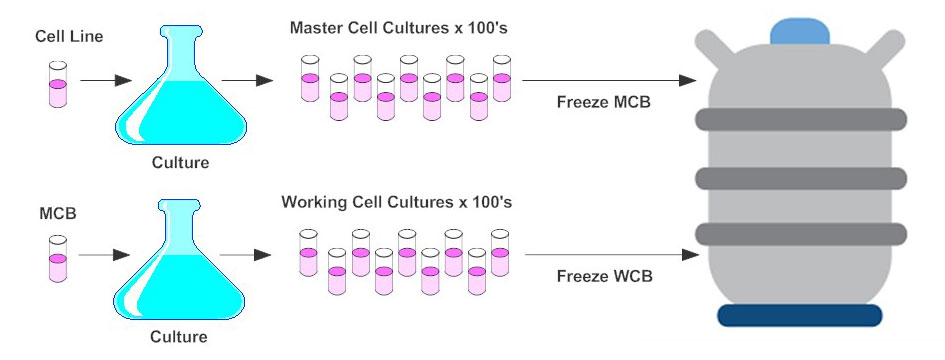
Recommendations for Cell Banks Used in GXP Assays - BioProcess InternationalBioProcess International
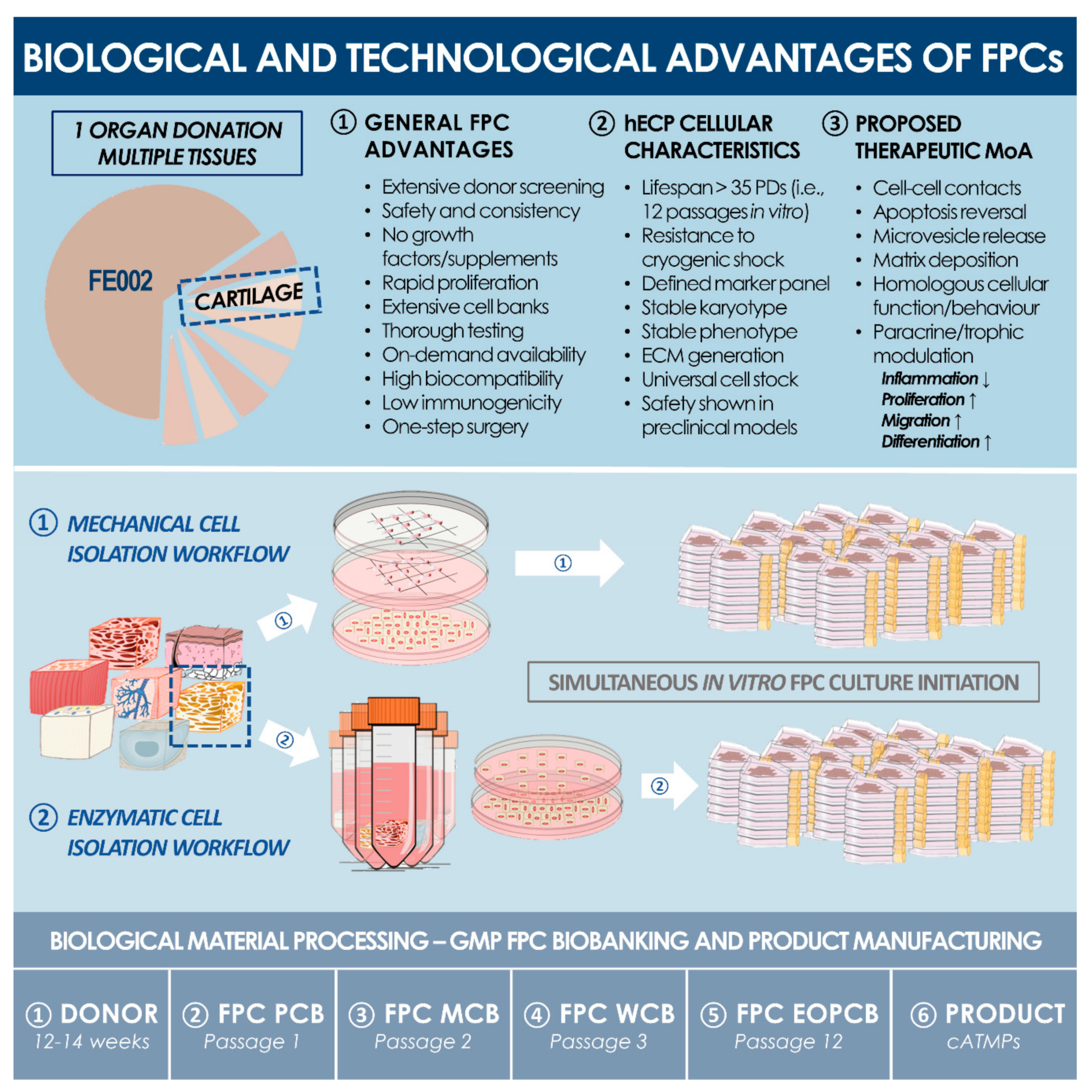
Biomolecules | Free Full-Text | Development of Standardized Fetal Progenitor Cell Therapy for Cartilage Regenerative Medicine: Industrial Transposition and Preliminary Safety in Xenogeneic Transplantation