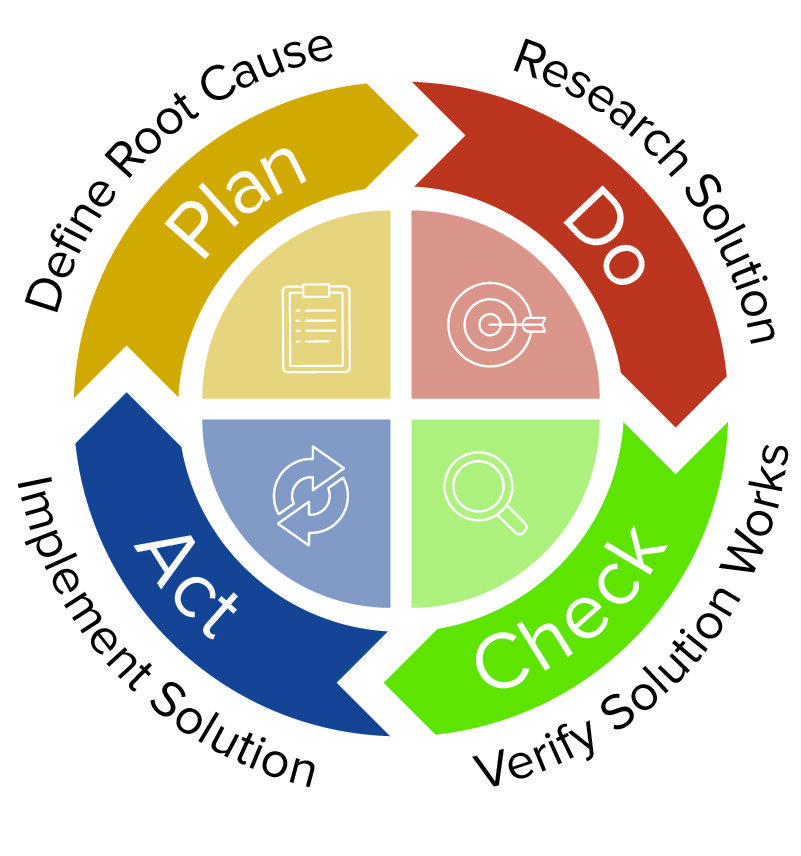
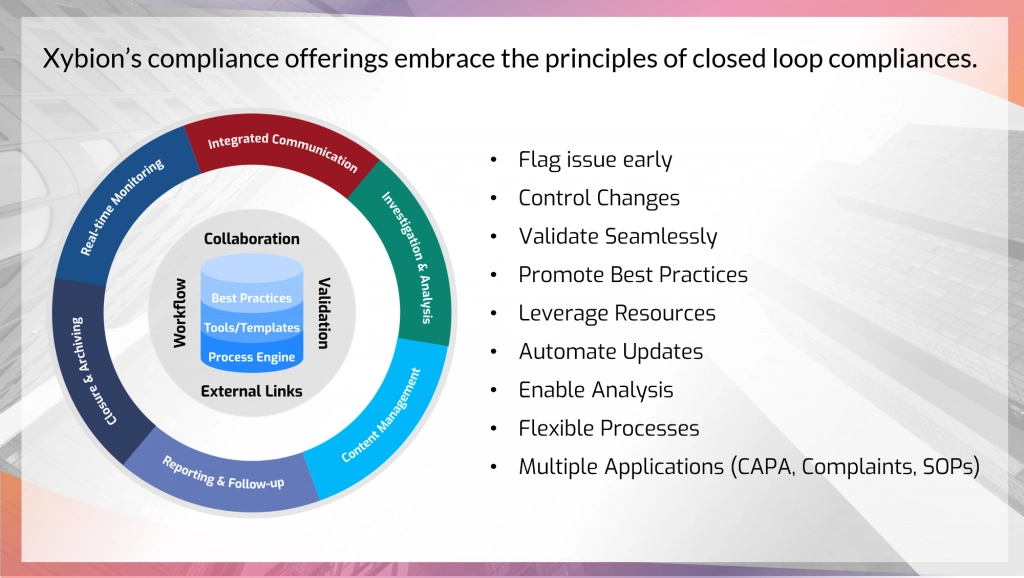
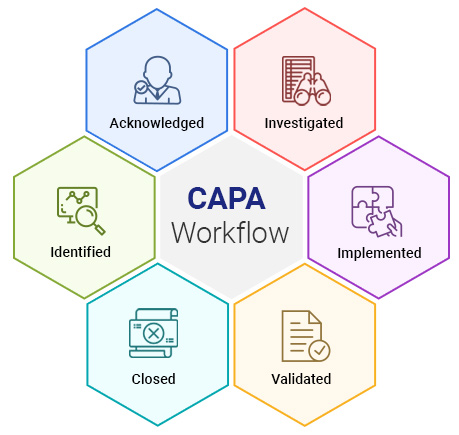
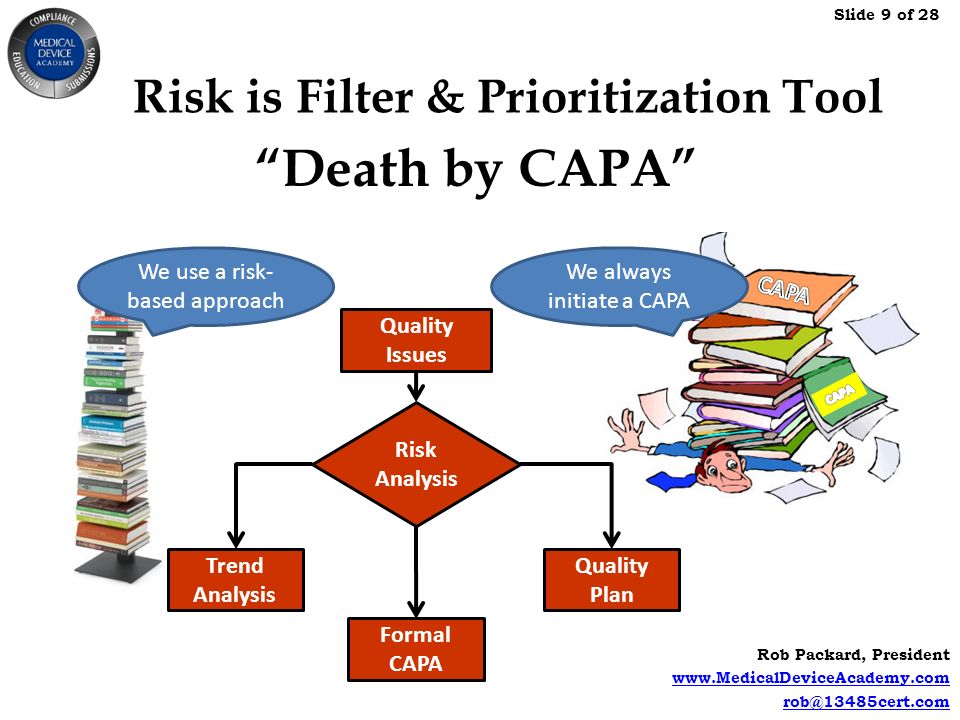
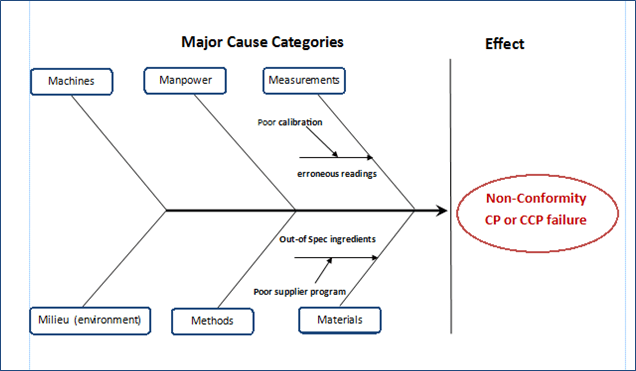
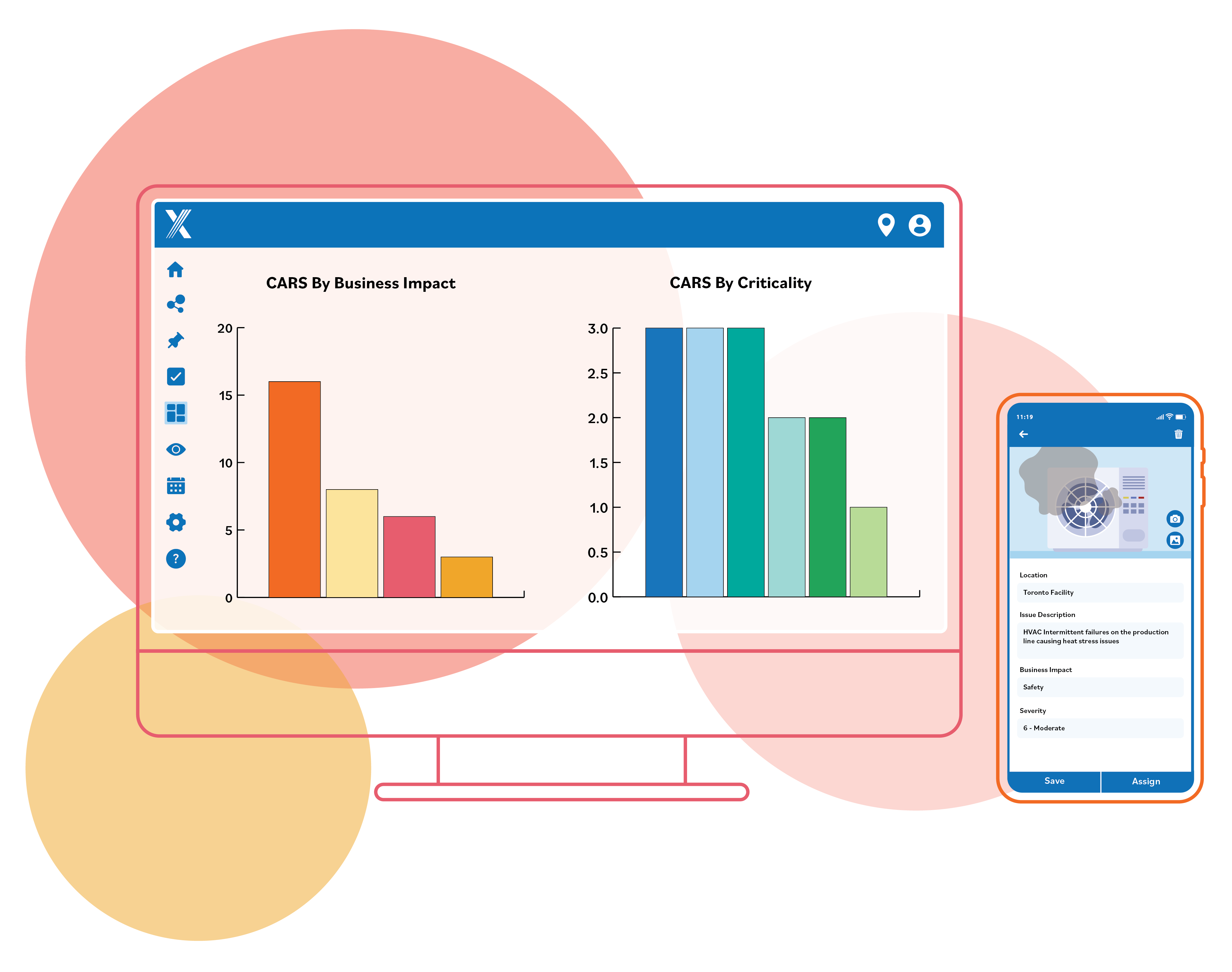
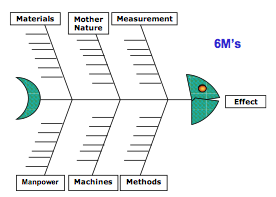
Gmpsop - Six step procedure for corrective and preventive action. Corrective and preventive action, typically named CAPA, is conducted at the implementation stage of most quality concern investigations in GMP environment. Corrective

Contextual Analysis for Practical Action (CAPA): a framework for understanding how context works and how to apply it for practical learning and implementing change in complex systems | Semantic Scholar

CAPA- Corrective Action & Preventive Action - knowledge-swami.com CAPA- Corrective Action & Preventive Action
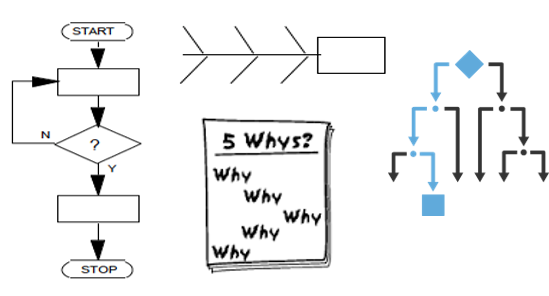
Creating a Problem-Solving Culture in Your Organization – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog