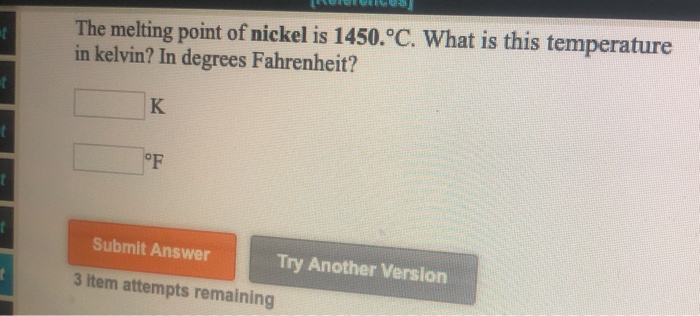
Temperature Temperature Scales Fahrenheit 212 o F 180 o F 32 o F Celcius 100 o C 0 o C Kelvin 373 K 100 K 273 K Boiling point of water Freezing point. - ppt download
How is aluminium melted at a lower temperature than its melting point (i.e. 660.3 degree Celsius) when mixed in small proportions to zinc? - Quora

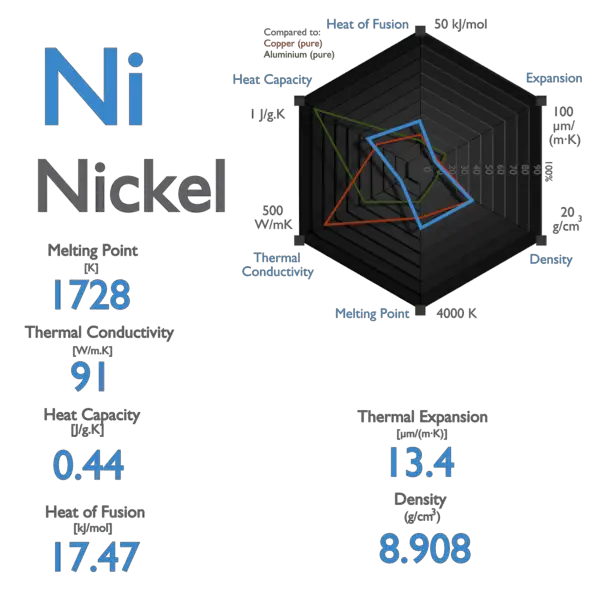
SOLVED: For nickel, Ni, the heat of fusion at its normal melting point of 1453 °C is 17.6 kJ/mol. The entropy change when 1.58 moles of liquid Ni freezes at 1453 °C,

SOLVED: 6.17. Estimate the melting point of nickel, Ni, if its AfusH is 17.61 kJ/mol and its AfusSis 10.21 Jmol-K (Compare this to a measured melting point of 1455*C )
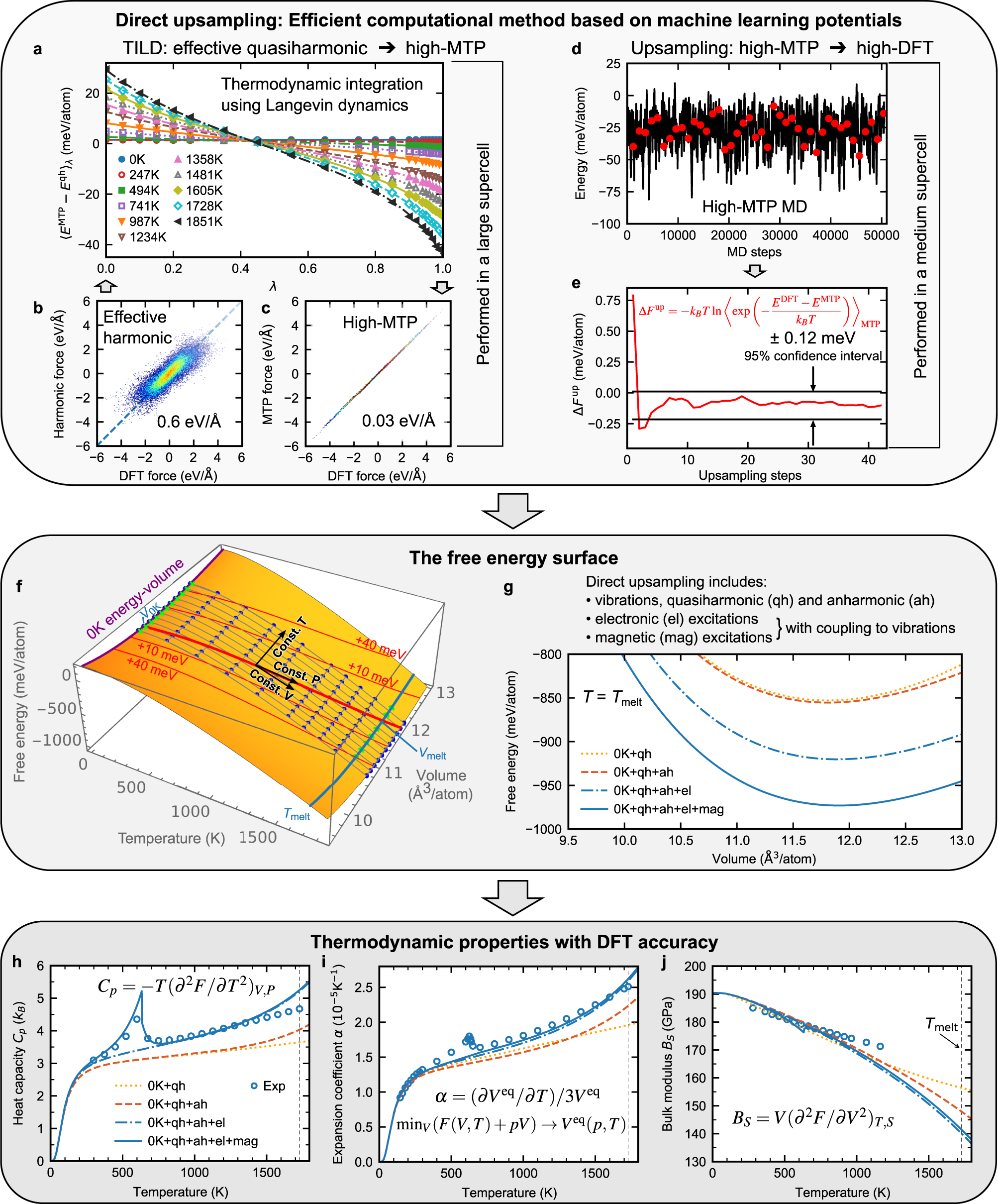
High-accuracy thermodynamic properties to the melting point from ab initio calculations aided by machine-learning potentials | npj Computational Materials