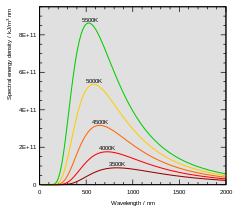
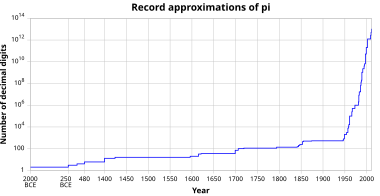
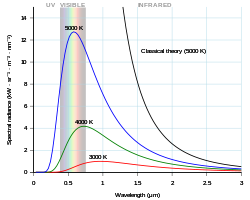
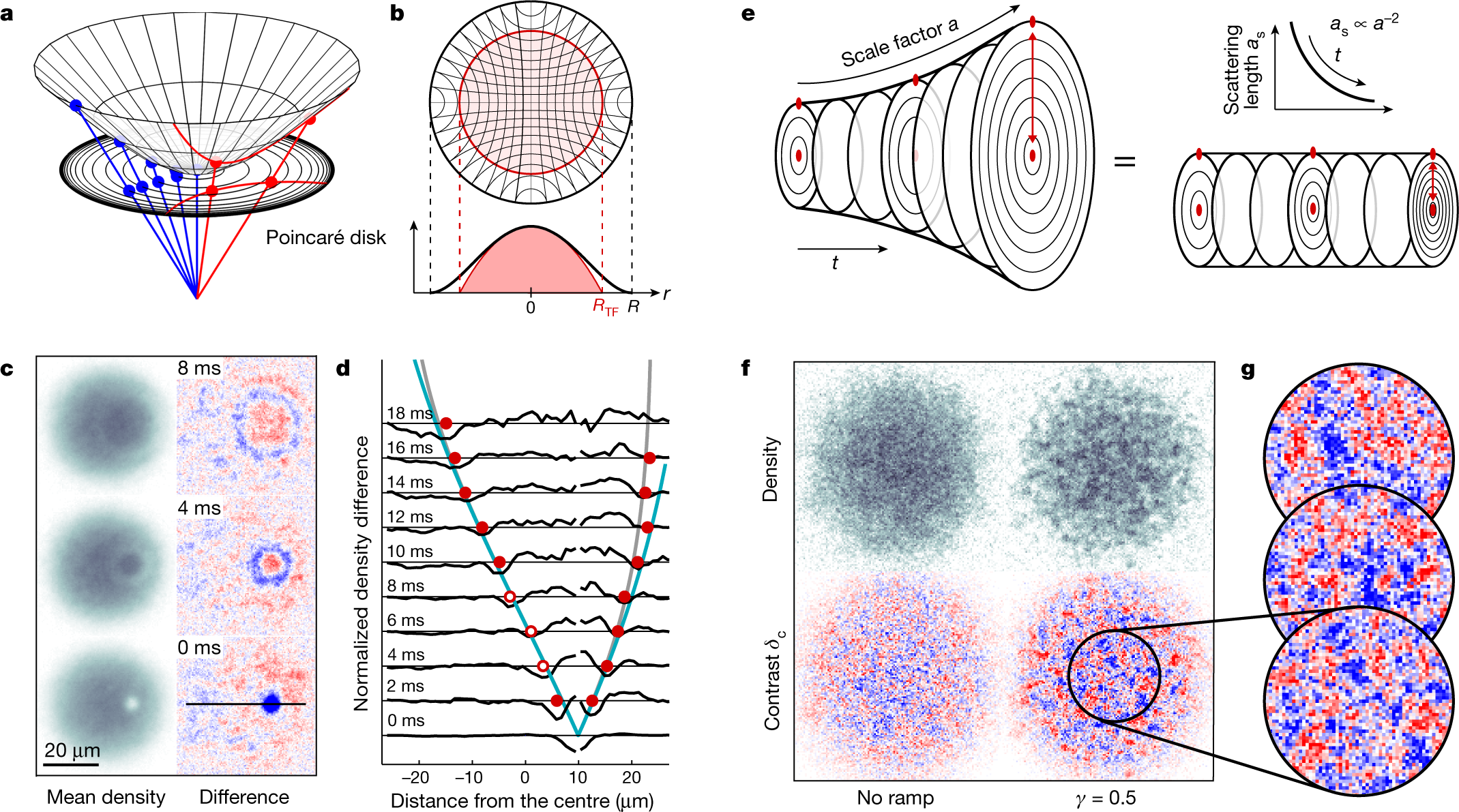
The value of Planck's constant is `6.63 xx 10^(-34)Js`. The speed of light is `3xx10^(17)nm s^ - YouTube
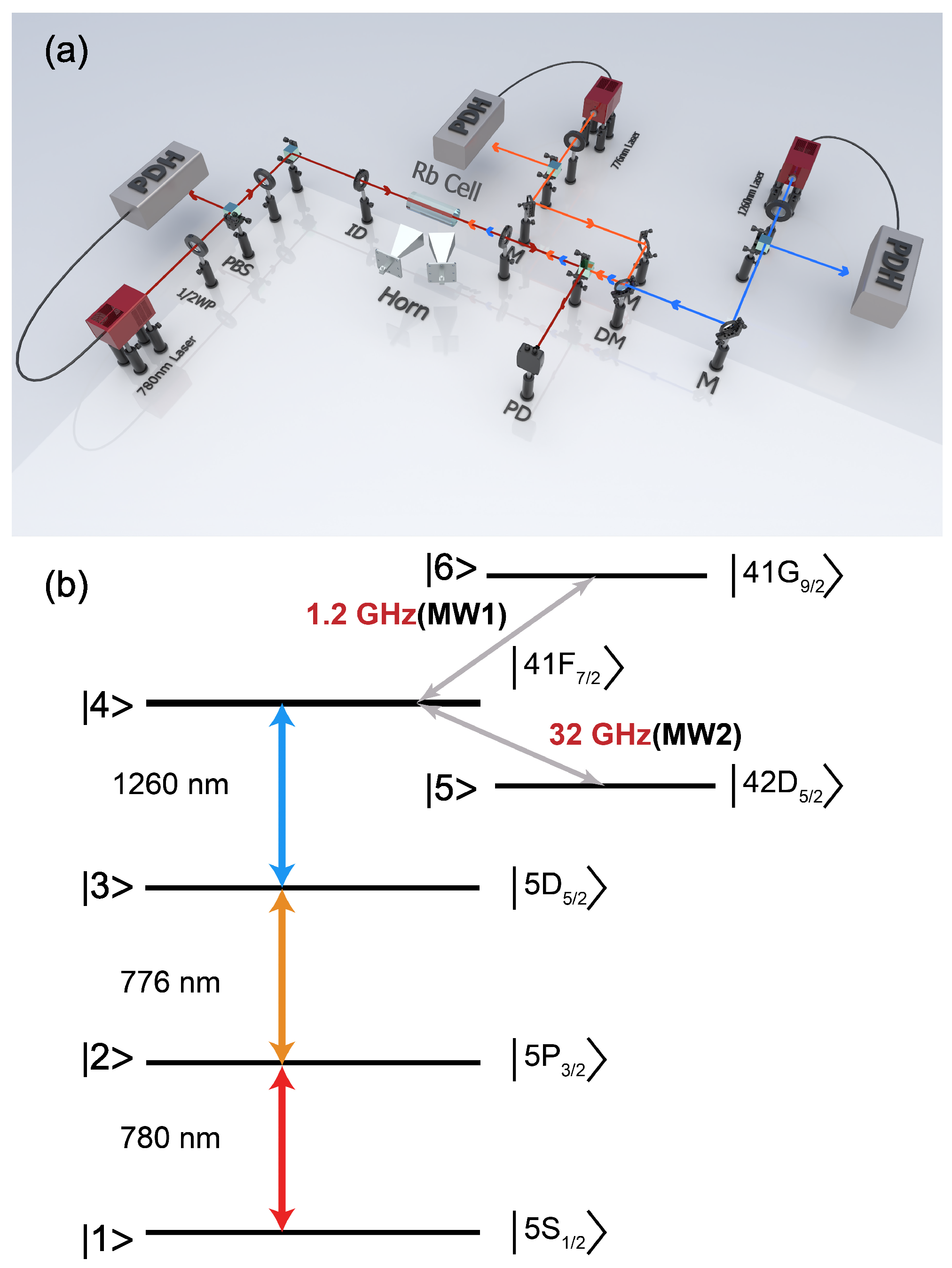
Photonics | Free Full-Text | Exclusive Effect in Rydberg Atom-Based Multi-Band Microwave Communication

Organic–inorganic hybrid perovskite scintillators for mixed field radiation detection - Xia - 2022 - InfoMat - Wiley Online Library

The value of Planck's constant (h) is 6.63 × 10^-34 Js . The velocity of light is 3.0 × 10^8 ms^-1 . Which value is closest to the wavelength (in meters) of

The value of Planck\'s constant is `6.63 xx 10^-34 Js`. The speed of light is `3 xx 10^(17)nms^(-1)` - YouTube
The value of Plank's constant is 6.63 × 10 34 Js. The speed of light is 3 × 1017 nm s 1 . Which value is closest with frequency of 6 × 1015 s 1 ?A. 25B. 75C. 10D. 50
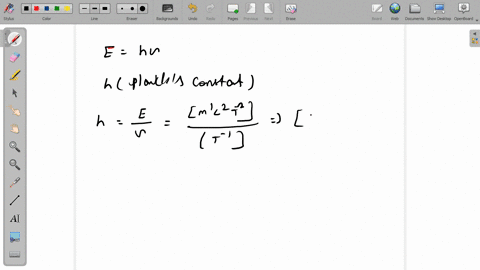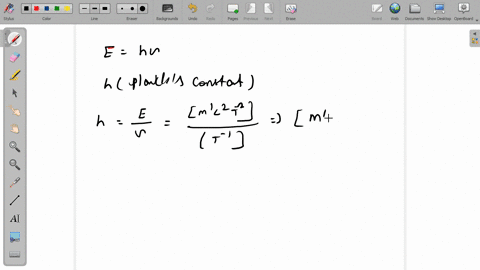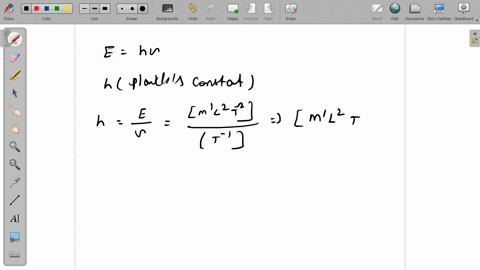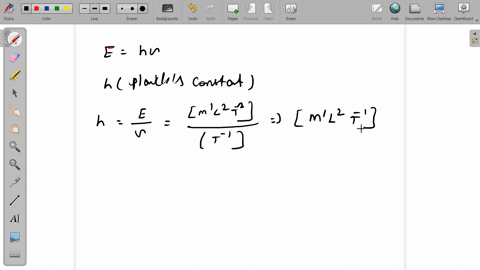
SOLVED:The dimensions of Planck's constant are the same as that of (a) linear impulse (b) work (c) linear momentum (d) angular momentum

The value of Planck's constant is 6.63 xx 10^(-34)Js. The speed of light is 3xx10^(17)nm s^(-1). Which value is the closed to the wavelength in nanometers of a quantum of light with
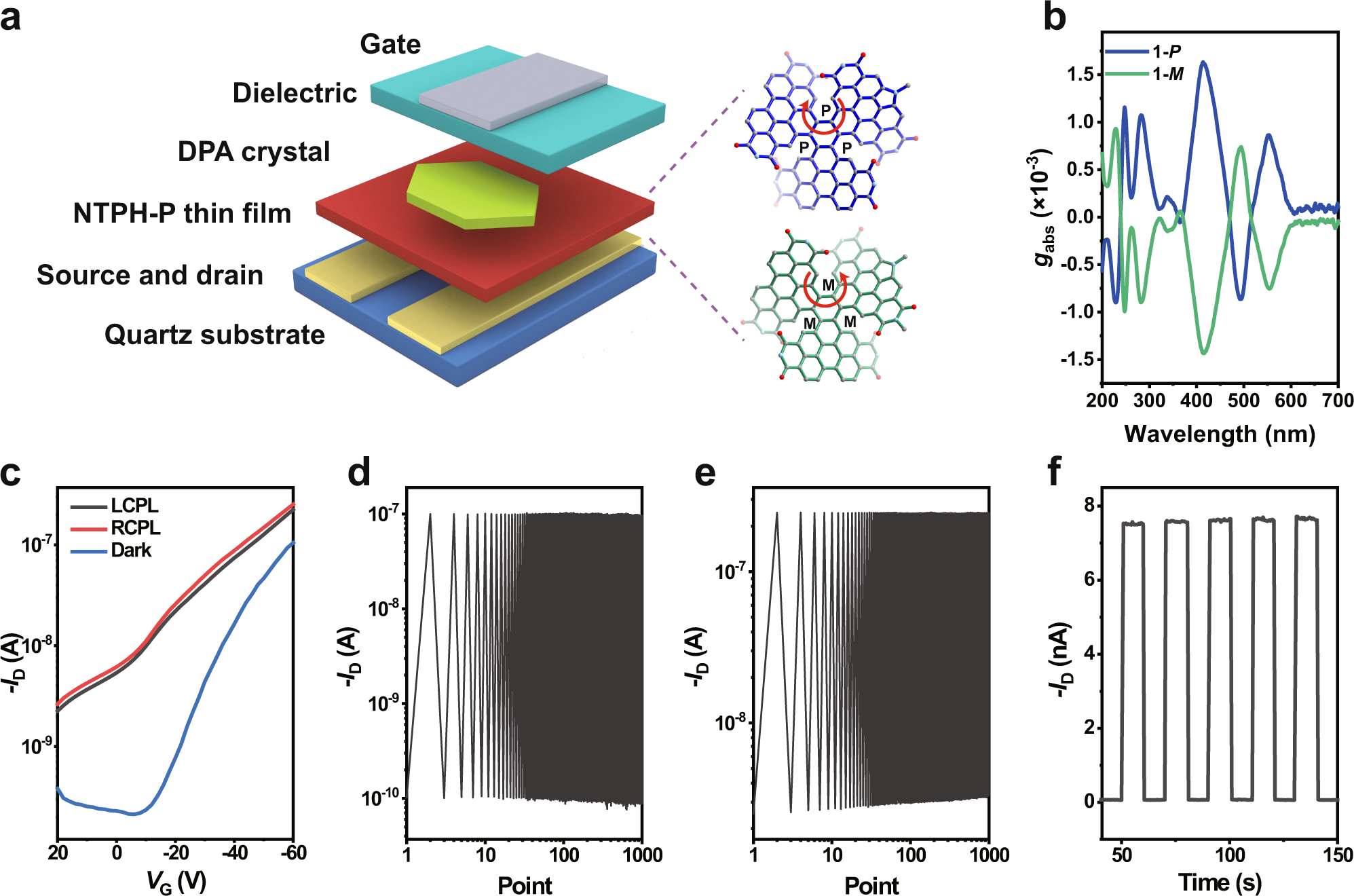
Organic donor-acceptor heterojunctions for high performance circularly polarized light detection | Nature Communications

Given: The mass of electron is 9.11 × 10^(–31)Kg Planck constant is 6.626 × 10^(–34)Js, the uncertainty involved in the measurement of velocity within a distance of 0.1Å is:-

The values of Planck's constant is 6.63 × 10^-34 Js. The velocity of light is 3.0 × 10^8 ms^-1 . Which value is closest to the wavelength in nanometres of a quantum of light which frequency of 8 × 10^15s^-1 ?

Symmetry | Free Full-Text | Position Dependent Planck's Constant in a Frequency-Conserving Schrödinger Equation